 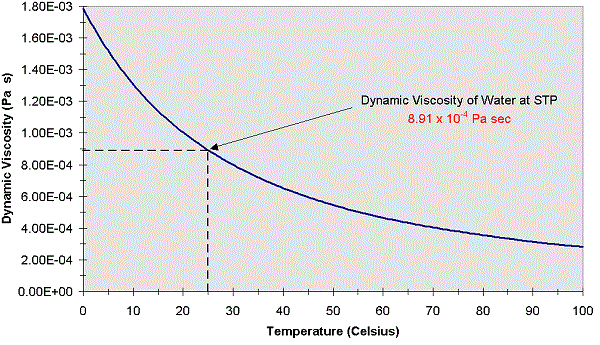
While this paper is addressed, principally, to fluid dynamicists these results should be of interest to physicists studying the liquid state and physical chemists interested in mixtures. Evidence from the specific heats of glycerol that the entropy of a glass exceeds that of a crystal at the absolute zero, J. This correlation can now be used at a given temperature to tailor make a mixture of prescribed kinematic viscosity. DH: 219.2: 293.15: Atalla, El-Sharkawy, et al., 1981: DH: 221.9: 298.15: Murthy and Subrahmanyam, 1977: DH. 14-18-Gives surface tension at 25 C for aqueous propylene glycols. The constants a, b and c are tabulated in the paper as functions of temperature. 14-12-Gives vapor viscosity of propylene glycols and glycerine from 0 C to 500 C. Heating of glycerin from room temperature to 100C decreases its viscosity by two orders of magnitude, thus allowing one to increase the Reynolds number by the. It is shown that all the data can be reasonably represented by the empirical correlation (ln ν m - ln ν w)/(ln ν g - ln ν w) = x g, where ν w, ν g and ν m are the kinematic viscosities of water, glycerol and the mixture respectively and x g is the mass fraction of glycerol in the mixture. Glycerin C3H8O3 CID 753 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety/hazards/toxicity information, supplier lists, and more. Apart from comprehensiveness and comparative accuracy the present measurements expose serious errors in the limited data that were earlier available on such mixtures. The measurements were made by using a series of Ubbelohde viscometers. We have measured the kinematic viscosity of glycerol-water mixtures, for glycerol mass fractions ranging from 0 to 1, in the temperature range 10-50 degrees C. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
